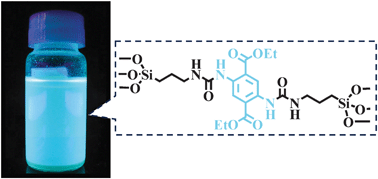
A bis-silylated diethyl 2,5-bis[N,N-(3-triethoxysilyl)propylurea]terephthalate precursor (5) was synthesized from a terephthalate derivative (3) and successfully used to prepare highly fluorescent organosilicas by hydrolytic self-condensation (DPM1) and co-condensation (DPM2) with tetraethoxysilane. The dyes 3 and 5 present absorption in the UV-Vis region and fluorescence emission in the yellow and blue regions, respectively. The red-shifted bands in the absorption spectra of dye 3 with respect to dye 5 can be related to the formation of intramolecular charge transfer (ICT) state even in the ground-state due to the strong electron-donor amino groups and moderate electron-withdrawing carbonyl groups present in the benzenic ring. The small Stokes' shift, as well as the high fluorescence quantum yield values discards the intramolecular proton transfer mechanism in the excited state. The fluorescence emission dependence on the solvent polarity indicates that an intramolecular charge transfer (ICT) or a locally excited (LE) state is taking place in the excited state to the dyes 3 and 5, respectively. The new fluorescent hybrid materials (DPM1 and DPM2) show absorption maxima in the solid state located at around 390 nm, indicating that the electronic structure of the fluorescent precursor was not significantly perturbed in the ground state after its self-condensation or co-condensation with TEOS. The fluorescent material DPM2 presents a photophysical behavior quite similar to the precursor in solution. On the other hand, the self-condensed material DPM1 presents a fluorescence emission maximum red-shifted by 33 nm, which can be probably attributed to π–π stacking between the terephthalate cores. Any evidence of auto-organization could be observed in these materials.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...