In situ chemical reduction of the Ta3N5 quantum dots coupled TaON hollow spheres heterojunction photocatalyst for water oxidation†
Abstract
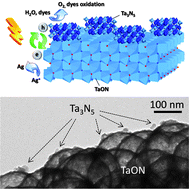
Photocatalytic oxygen evolution with a high efficiency was achieved using tantalum nitride (Ta3N5) quantum dots (QDs) coupled TaON hollow spheres (Ta3N5–TaON). TaON hollow spheres coupled with the surface enriched Ta3N5 QDs were prepared by an in situ chemical reduction route in ammonia solution at −45 °C and were characterized by X-ray diffraction, scanning electron microscopy, transmission electron microscopy, X-ray photoelectron spectra, UV-vis diffuse reflectance spectra and photoluminescence spectra. The Ta3N5–TaON composites containing 4 mol% Ta3N5 QDs showed a high rate of O2 production at 208.2 μmol h−1 with an apparent quantum efficiency of 67% under 420 nm light. The rate of oxygen formation of the Ta3N5–TaON heterojunction was 3.3 times higher than that of the pristine TaON hollow spheres. Furthermore, relative photoelectrochemical properties of Ta3N5–TaON composite photoelectrodes were investigated. The resulting 4 mol% Ta3N5–TaON heterojunction films exhibited a photocurrent of ca. 2.7 mA cm−2 under visible light irradiation at 1.0 V vs. SCE in Na2SO4 solution. This excellent photocatalytic activity is ascribed to the Ta3N5 QDs that alter the energy levels of the conduction and valence bands in the coupled semiconductor system and the slow recombination of photogenerated electron–hole pairs. Moreover, the Ta3N5–TaON composite exhibited strong durability which could be attributed to the inhibition of Ta3N5 QDs leaching owing to its strong interaction with TaON.

 Please wait while we load your content...
Please wait while we load your content...