Synthesis and characteristics of novel layered silicates HUS-2 and HUS-3 derived from a SiO2–choline hydroxide–NaOH–H2O system†
Abstract
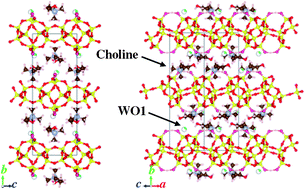
Three kinds of layered silicates, HUS-2, HUS-3, and HUS-4, were synthesized using amorphous silica, sodium hydroxide, and choline hydroxide as a structure-directing agent (SDA). Structural analyses by powder X-ray diffractometry and solid-state magic-angle-spinning (MAS) NMR spectroscopy revealed that HUS-2 had a layered structure, which was composed of four-, five-, and six-membered rings. Its framework topology was partially similar to that of a HEU-type zeolite including bre (10T)-type composite building units. Structural refinement by the Rietveld method showed that the choline cations that were used as an SDA were incorporated into the interlayers. Double silicate layers were included in each unit cell. The interlayer distance was estimated to be ca. 0.36 nm. The presence of hydrogen bonding between adjacent terminal O atoms was clearly revealed by 1H MAS NMR spectroscopy and through the electron-density distribution obtained by the maximum entropy method. Although the crystal structure of the layered silicate HUS-3 could not be analysed, the HUS-3 was transformed into a CDO-type zeolite by calcination, indicating that the structure of HUS-3 was partially similar to that of a CDO-type zeolite precursor. The crystal structure of HUS-4 was similar to that of PLS-1 synthesized using tetramethylammonium hydroxide. We also demonstrated that the layered silicates HUS-2 and HUS-3 exhibited high catalytic activities as base catalysts in a Knoevenagel condensation reaction.

 Please wait while we load your content...
Please wait while we load your content...