Understanding the high catalytic activity of propylsulfonic acid-functionalized periodic mesoporous benzenesilicas by high-resolution 1H solid-state NMR spectroscopy†
Abstract
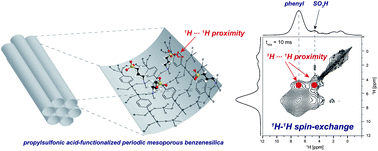
Propylsulfonic acid-functionalized periodic mesoporous benzenesilica (Ph-PMO-SO3H, 1) has been shown to be exceptional solid

 Please wait while we load your content...
Please wait while we load your content...