The focus of this study is on the identification of precursors in solution that might act
as building blocks when solid uranyl(VI) poly-peroxometallate clusters
containing peroxide and hydroxide bridges are formed. The
precursors could be identified by using carbonate as an auxiliary ligand that prevented the formation of large clusters, such as the ones found
in solids of fullerene type. Using data from
potentiometric and NMR (17O and 13C)
experiments we identified the following complexes and determined their equilibrium
constants:
(UO2)2(O2)(CO3)46−,
UO2(O2)CO32−,
UO2(O2)(CO3)24−,
(UO2)2(O2)(CO3)22−,
(UO2)2(O2)2(CO3)2− and
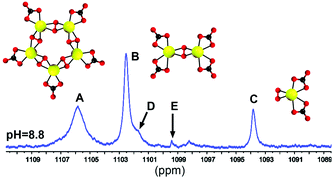
[UO2(O2)(CO3)]510−. The NMR spectra of the pentamer show that all uranyl and carbonate
sites are equivalent, which is only consistent with a ring structure built from uranyl
units linked by peroxide bridges with the carbonate
coordinated “outside” the ring; this proposed structure is very similar to
[UO2(O2)(oxalate)]510− identified by Burns
et al. (J. Am. Chem. Soc., 2009, 131, 16648; Inorg.
Chem., 2012, 51, 2403) in
K10[UO2(O2)(oxalate)]5·(H2O)13;
similar ring structures where oxalate or carbonate has been replaced by hydroxide are
important structure elements in solid poly-peroxometallate complexes. The equivalent
uranyl sites in
(UO2)2(O2)2(CO3)2−
suggest that the uranyl-units are linked by the carbonate
ion and not by peroxide.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...