Chelation-controlled molecular morphology: aminal to imine rearrangements†
Abstract
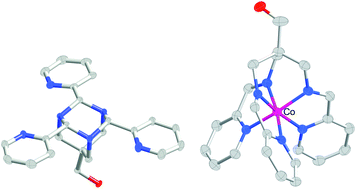
Reactions between the tripodal hydroxytriamine, 2,2-bis(aminomethyl)-3-aminopropan-1-ol, “hytame”, and the isomeric pyridine aldehydes generate in all cases the tris(aminal) species based on a 1,3,5-triaza-adamantane skeleton. In all cases also, the product from water under basic conditions consists of an approximately 1 : 9 mixture of the triequatorial and monoaxial–diequatorial isomers. While all these tripyridyltriaza-adamantanes appear capable of acting as Lewis bases, in particular cases metal ion binding leads to a radical structural rearrangement. These cases involve the pyridine-2-aldehyde derivatives only and certain transition metal ions (notably Fe(II)), and result in the conversion of the tris(aminal) into its isomeric tris(imine) form. This is apparently favoured because it can act as a hexadentate ligand towards a single metal ion, although kinetic influences are clearly important in this chemistry because template reactions of the triamine, pyridine-2-aldehyde and several metal ions give much better yields of the tris(imine) complex than do analogous rearrangement reactions. For the low-spin, kinetically inert Fe(II) complex of the tris(imine), its formation is apparently so favourable that it is generated via aldehyde unit exchange when the aza-adamantanes derived from pyridine-3- and -4-aldehyde are heated with a mixture of Fe(II) and pyridine-2-aldehyde. When the kinetically labile Zn(II) complex is treated with EDTA, the metal ion is extracted but the released ligand does not undergo valence tautomerisation to what would be expected to be the triaxial isomer of the tripyridyltriaza-adamantane but instead rapidly undergoes partial hydrolysis before slowly forming the mixture of triequatorial and monoaxial–diequatorial isomers.

 Please wait while we load your content...
Please wait while we load your content...