A family of hexa-coordinated ruthenium(II) complexes of bis(N-pyridylimidazolylidenyl)methane (L) were prepared and structurally characterized. Carbene transfer reactions of [Ru(p-cymene)Cl2]2, [Ru(CO)2Cl2]n and RuHCl(CO)(PPh3)3 with silver–NHC complexes in situ generated from [H2L](PF6)2 and Ag2O afforded [RuL(CH3CN)2](PF6)2 (1), [Ru2L(p-cymene)2Cl2](PF6)2 (2), [RuL(CO)2](PF6)2 (3) and [RuL(PPh3)2](PF6)2 (4), respectively. The reactions of 1 towards several N- and P-donors were studied. The treatment of 1 with 1,10-phenanthroline resulted in the substitution of one pyridine and one acetonitrile molecule affording [RuL(phen)(CH3CN)](PF6)2 (5) as a mixture of two isomers. Reaction of 1,2-bis(diphenylphosphino)ethane (dppe) and 1 gave [RuL(dppe)(CH3CN)2](PF6)2 (7), in which two pyridines were substituted by a dppe ligand trans to two NHC groups. In contrast, reactions of 1 with ethane-1,2-diamine, propane-1,3-diamine and 3,5-dimethyl-1H-pyrazole led to the substitution of acetonitrile and subsequent N–H addition of the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N bond of the coordinated acetonitrile yielding [RuL(ethane-1,2-diamine)(N-(2-aminoethyl)acetimidamide)](PF6)2 (8), [RuL(propane-1,3-diamine)(N-(3-aminopropyl)acetimidamide)](PF6)2 (9) and RuL(1-(3,5-dimethyl-1H-pyrazol-1-yl)ethanimine)(CH3CN)](PF6)2 (10), respectively.
N bond of the coordinated acetonitrile yielding [RuL(ethane-1,2-diamine)(N-(2-aminoethyl)acetimidamide)](PF6)2 (8), [RuL(propane-1,3-diamine)(N-(3-aminopropyl)acetimidamide)](PF6)2 (9) and RuL(1-(3,5-dimethyl-1H-pyrazol-1-yl)ethanimine)(CH3CN)](PF6)2 (10), respectively.
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N bond of the coordinated acetonitrile yielding [RuL(ethane-1,2-diamine)(N-(2-aminoethyl)acetimidamide)](PF6)2 (8), [RuL(propane-1,3-diamine)(N-(3-aminopropyl)acetimidamide)](PF6)2 (9) and RuL(1-(3,5-dimethyl-1H-pyrazol-1-yl)ethanimine)(CH3CN)](PF6)2 (10), respectively.
N bond of the coordinated acetonitrile yielding [RuL(ethane-1,2-diamine)(N-(2-aminoethyl)acetimidamide)](PF6)2 (8), [RuL(propane-1,3-diamine)(N-(3-aminopropyl)acetimidamide)](PF6)2 (9) and RuL(1-(3,5-dimethyl-1H-pyrazol-1-yl)ethanimine)(CH3CN)](PF6)2 (10), respectively.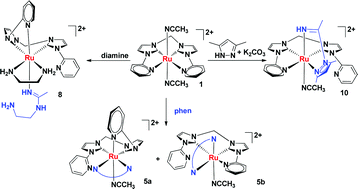

 Please wait while we load your content...
Please wait while we load your content...