High surface area TiO2–ZrO2 prepared by caustic solution treatment, and its catalytic efficiency in the oxidehydrogenation of para-ethyltoluene by CO2
Abstract
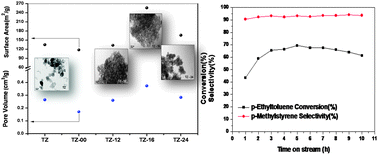
High surface area TiO2–ZrO2 (1 : 1) has been synthesized by co-precipitation—digestion method using KOH solution as a hydrolyzing agent as well as a digestion medium. The quenching of the mother liquor solution at elevated temperatures resulted in the amorphization of the TiO2–ZrO2 mixed

 Please wait while we load your content...
Please wait while we load your content...