Enhanced electronic conductivity by controlled self-doping in pyrochlores
Abstract
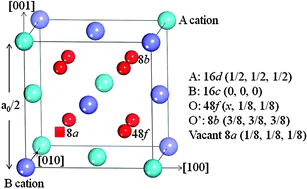
Most 5d transition-metal (TM) pyrochlores exhibit metallic behavior, but 3d and 4d TM pyrochlores are generally electronic semiconductors or insulators. Here, we report a semiconductor–metal transition induced by introducing excess Ti metal as interstitials into Y2Ti2O7. These Ti interstitials prefer anion vacant 8a sites or bridge sites between two neighboring

 Please wait while we load your content...
Please wait while we load your content...