The effect of metal cations on the nature of the first electronic transition of liquid water as studied by attenuated total reflection far-ultraviolet spectroscopy
Abstract
The first electronic transition (Ã ← ![[X with combining tilde]](https://www.rsc.org/images/entities/i_char_0058_0303.gif) ) of liquid
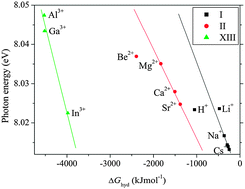
) of liquid ![[X with combining tilde]](https://www.rsc.org/images/entities/i_char_0058_0303.gif) transition energies of 1 M electrolyte solutions are higher (Li+: 8.024 eV and Cs+: 8.013 eV) than that of pure
transition energies of 1 M electrolyte solutions are higher (Li+: 8.024 eV and Cs+: 8.013 eV) than that of pure ![[X with combining tilde]](https://www.rsc.org/images/entities/i_char_0058_0303.gif) transition energies are mostly attributable to the hydrogen bond formation energies of
transition energies are mostly attributable to the hydrogen bond formation energies of ![[X with combining tilde]](https://www.rsc.org/images/entities/i_char_0058_0303.gif) transition energies of the water–cation complexes depend on the hydration structures of the
transition energies of the water–cation complexes depend on the hydration structures of the ![[X with combining tilde]](https://www.rsc.org/images/entities/i_char_0058_0303.gif) transition energies of the
transition energies of the ![[X with combining tilde]](https://www.rsc.org/images/entities/i_char_0058_0303.gif) transition bands measured by ATR-FUV
transition bands measured by ATR-FUV

 Please wait while we load your content...
Please wait while we load your content...