Stereodynamics of multistate roaming
Abstract
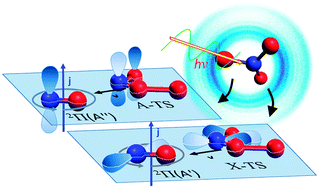
We present a molecular level description of NO3 → NO + O2 photodissociation for both of the experimentally observed reaction pathways using the results of ion imaging experiments and recent theoretical studies. Vector correlation and Λ doublet propensity measurements have been performed on state-selected NO fragments in order to further characterize the stereodynamics of this reaction. Previous measurements (Grubb et al., Science, 2012, 1075–1078) of relative Λ doublet propensities along with ab initio calculations revealed that both pathways arise from roaming-type mechanisms, but each pathway arises from roaming on a different electronic potential. This model, however, assumes that NO3 dissociation takes place in the molecular plane. In the present paper, we have confirmed this assumption through speed-dependent vector correlation measurements. Strong perpendicular correlations between the velocity vector v and the angular momentum vector j are observed in the NO fragment originating from both pathways, in agreement with a constrained planar dissociation. These results are discussed in light of the absence of vector correlations in other roaming systems, which have previously been characterized by an unconstrained intra-molecular abstraction. We show that geometrical constraints should in fact be quite prevalent in roaming dynamics, and are analogous to the geometrical constraints of the corresponding bimolecular abstraction reaction.

 Please wait while we load your content...
Please wait while we load your content...