Surface pressure induced 2D-crystallization of POM-based surfactants: preparation of nanostructured thin films†
Abstract
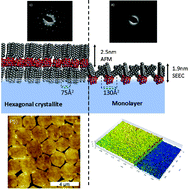
The present study investigates the deposition of hybrid organic/inorganic monolayers composed of polyoxometalate (POM) of the Keggin-type [PW12O40]3− electrostatically linked to three alkylammonium surfactants. The surface pressure dependence, i.e. Langmuir isotherm, as well as their 2D self-assembly has been investigated with mono- and double-chained surfactants and by varying the alkyl chain length. Stable monolayers were obtained for the hexadecyltrimethylammonium and the didodecyldimethylammonium POM surfactants, called respectively C16POM and 2C12POM. It was found that increasing the surface pressure led to the formation of crystallized nano- and micro-domains in coexistence with the monolayer. The crystallites formed were monodisperse in their size repartition and showed various shapes and sizes: oblate nanoparticles of nanometric size (height ca. 7 nm, diameter ca. 140 nm) for C16POM and micrometric 2D-hexagonal crystallites for 2C12POM. The monolayers and the micro-(nano-) crystallites have been characterized after transfer on silicon wafers by means of atomic force microscopy (AFM), transmission electron microscopy (TEM) and surface-enhanced ellipsometric contrast (SEEC). High crystallinity of the hexagonal aggregates was revealed by electron diffraction (ED). For 2C12POM the monolayer formed before crystallisation is in a 2D-liquid state with the alkyl chains pointing out of the water with an average angle of the alkyl chains to the normal of the plane estimated at 50°. It is shown that the surface pressure induced crystallization of the POM surfactant results in the change of the ammonium surfactant arrangement around the POM with the alkyl chains pointing up and down the POM units showing a reverse bilayer-like structure. This change in the ammonium surfactant arrangement in response to the increase of the surface pressure enables (i) to reduce the molecular area and (ii) the formation of a 2D-hexagonal crystalline lattice of POM units. On the other hand for C16POM the formation of oblate nanoparticles is driven by a three-dimensional growth at the water surface.

 Please wait while we load your content...
Please wait while we load your content...