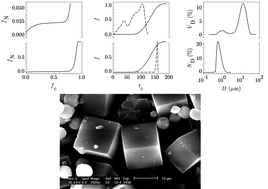
Influence of the simultaneous change of batch alkalinity (A = [Na2O/H2O]b) and total batch silica concentration (tsc = [SiO2/H2O]b) of the reaction mixture, at two characteristic batch molar ratios, y = [SiO2/Al2O3]b (1.3 and 2.0), on the kinetics of nucleation, crystal growth and overall crystallization and on the particulate properties of crystallized zeolite A was investigated. It was found that the mentioned processes during crystallization in high and medium alkaline systems (0.0165 ≤ A ≤ 0.046) take place in accordance with previously described experimental evidence, but that the anomalous kinetics of nucleation during crystallization of zeolite A from low alkaline systems (A ≤ 0.01) was not either predicted theoretically or, until recently, evidenced experimentally. Results of the analysis of distribution of nuclei in gel matrix have shown that in low alkaline systems, most of the nuclei are concentrated in the “centers” (“cores”) of gel particles and the rest of the nuclei are distributed between the “surfaces” and “cores” of gel particles. Population balance analyses of the crystallization processes showed that the nucleation in the low alkaline systems takes place in two steps as a consequence of the mentioned distribution of nuclei in the gel matrix. Another consequence of the anomalous distribution of nuclei in the gel matrix is that more than 95 wt% of zeolite A is formed by the growth of the nuclei released from the gel matrix during the first step of nucleation, and thus the maximum rate of nucleation in the second step is reached at the crystallization time when about 80–90 wt% of gel has been transformed into zeolite A. This is, at the same time, the reason that the crystal size distributions of the products obtained by crystallization of zeolite A from low-alkaline systems are characterized by two separate crystal populations.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...