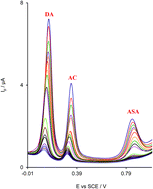
This paper reports the study of the electrochemical oxidation of dopamine (DA), acetaminophen (AC), and aspirin (ASA) comparatively conducted at a ruthenium oxide nanoparticles modified glassy carbon electrode (RuON-GCE) and at a bare glassy carbon electrode (BGCE). The obtained results show that the reversibility of DA, AC and ASA is significantly improved on RuON-GCE in comparison with BGCE. Using differential pulse voltammetry, the prepared sensor showed good sensitivity and selectivity for the individual and the simultaneous determination of DA, AC and ASA. The effect of the modification of the electrode surface on the diffusion coefficient, D, of DA, AC, and ASA in solution as well as the heterogeneous electron transfer rate constant, k′, between the three species and both electrode (BGCE and RuON-GCE) surfaces are studied. The results indicate that the D values of the three species are independent but their k′ values are dependent on the nature of the electrode surface.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...