Immobilization of trypsin on graphene oxide for microwave-assisted on-plate proteolysis combined with MALDI-MS analysis†
Abstract
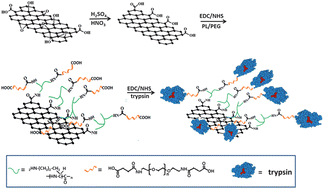
With an ultra-high surface area and abundant functional groups, graphene oxide (GO) provides an ideal substrate for the immobilization of trypsin. We demonstrated that trypsin could be immobilized on GO sheets assisted by

 Please wait while we load your content...
Please wait while we load your content...