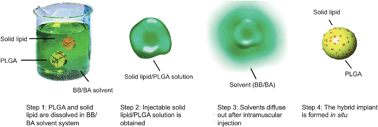
Effective sustained delivery systems are in urgent demand for patients receiving long-term medication (e.g., for schizophrenia), as poor compliance often leads to unproductive treatments. Although the conventional PLGA-based implants have been extensively studied for sustained delivery, they require invasive surgical operations. A more challenging problem is the burst release effect that is characteristic of these systems. Presented here is a novel, hybrid PLGA injectable implant for long-acting risperidone for improved antipsychotic treatment, avoiding pitfalls of the oral form such as poor bioavailability, short half-life and wide fluctuation of blood drug concentrations. This hybrid solid lipid/PLGA implant (S/P-I) is formed in situ following intramuscular injection. The unique advantages of incorporating solid lipid are the improved drug release profiles and reduced burst effect by altering the morphology and phase inversion of the implant, as well as the improved injectability. This hybrid system shows the improved in vitro sustained drug release profiles, significantly reducing the burst effect. In vivo studies show the prolonged mean residence time (MRT) of 86.8 h for S/P-I, compared with 5.8 h for the risperidone solution and 32.6 h for the PLGA implant. Notably, S/P-I also displays a relatively high bioavailability at 87.8%, indicating nearly complete drug release, which is not typical in conventional PLGA-based sustained drug delivery systems. The in vivo S/P-I underwent almost complete biodegradation, with less than 2% remaining after 28 days. The findings provide a simple, yet useful and effective solution for controlling the burst effect in PLGA implant systems with glyceryl monostearate as controlled release component. S/P-I is a promising system for long-acting risperidone for improved antipsychotic treatment.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...