Monte Carlo simulations of the electrical double layer forces in the presence of divalent electrolyte solutions: effect of the ion size†
Abstract
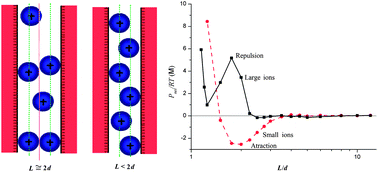
In this paper, the effect of ion size on the mean forces between two charged plates in the presence of divalent counterions is analyzed with the help of Monte Carlo simulations in the framework of the primitive model. Inspired by a preliminary work in which a particular and isolated case was presented, we propose a more systematic survey considering both like and oppositely charged plates, different surface charge densities (with magnitudes ranging from 0.01 to 0.2 C m−2) and two very different ionic strengths (0.5 and 500 mM). The effect of ion size is probed comparing systematically results for the most commonly used ionic diameter (0.425 nm) and greater values reported in the scientific literature for hydrated ions. These greater values were previously and successfully employed in other areas (e.g., electrokinetic behaviour in the presence of divalent and trivalent ions). Our simulations show that force–distance profiles strongly depend on the ion size. Consequently, some of the ‘classical’ findings obtained from simulations must be carefully reconsidered. In particular, the widely known and reported attraction between like-charged plates (or macroions) becomes repulsion with increasing ion size.
- This article is part of the themed collection: International Soft Matter Conference 2010

 Please wait while we load your content...
Please wait while we load your content...