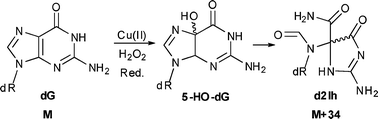
Reactive oxygen species attack both base and sugar moieties in DNA with a preference among the bases for reaction at guanine. In the present study, 2′-deoxyguanosine (dG) was oxidized by a copper-mediated Fenton reaction with the reductants ascorbate or N-acetyl-cysteine, yielding oxidation on both the base and the sugar. The primary oxidized lesions observed in these studies include the 2′-deoxyribonucleosides of 8-oxo-7,8-dihydroguanosine (dOG), spiroiminodihydantoin (dSp), guanidinohydantoin (dGh), oxazolone (dZ), and 5-carboxamido-5-formamido-2-iminohydantoin (d2Ih), as well as the free base guanine. d2Ih was the major product observed in the nucleoside, single- and double-stranded oligodeoxynucleotide contexts and is proposed to arise from oxidation at C5 of guanine. Product distribution studies provide insight into the role of the reductant in partitioning of dG base oxidation along the C5 and C8 pathways.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...