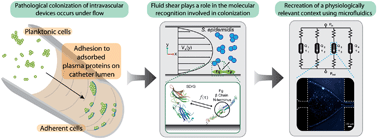
Staphylococcus epidermidis is an opportunistic pathogen that has been implicated in hospital-acquired infections, specifically related to implanted intravascular devices. S. epidermidis adhesion is a mechanism of colonization, leading to pathogenesis. Here we demonstrate an easily fabricated and robust parallel microfluidic platform to investigate the physiologically-relevant effects of fluid shear on S. epidermidis adhesion to human fibrinogen (hFg) with increased experimental throughput. In situ molecular patterning using fluid flow boundaries allows for isolation of the molecular interactions in highly defined shear stress environments, while keeping the device operation simple and reproducible. We characterize two modes of attachment of S. epidermidis to hFg coated surfaces. Single colonies adhere in high fractions at low shear stresses (~1 dyne cm−2) and adhesion decays with increasing shear. However, clusters of bacteria adhere the highest at median wall shear stress (up to 10 dyne cm−2), and adhesion subsequently decays above this critical shear stress. This initial characterization suggests a previously unobserved phenomenon of shear activated cell–cell adhesion in S. epidermidis, which acts to increase the overall attachment strength to hFg. Both of these modes of attachment are dependant upon the presence of intact hFg, indicating that adhesion is resultant from specific molecular recognition between the bacteria and human fibrinogen. This platform provides new insight into complex host–pathogen interactions, and will allow for further investigation of colonization and pathogenesis in more physiologically relevant conditions.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...