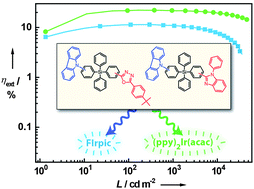
A series of carbazole-containing tetraarylsilane compounds, namely p-BISiPCz (1), m-BISiPCz (2), p-OXDSiPCz (3) and m-OXDSiPCz (4) were designed and synthesized by incorporating electron-donating carbazole and electron-accepting benzimidazole or oxadiazole into one molecule via a silicon-bridge linkage mode. Their thermal, photophysical and electrochemical properties can be finely tuned through the different groups and linking topologies. The di-para-position compounds 1 and 3 display higher glass transition temperatures and slightly lower triplet energies than their di-meta-position isomers 2 and 4, respectively. The four compounds exhibit similar HOMO levels (5.60–5.63 eV), while the LUMO level of 3 (2.36 eV) is slightly lower than that of 4 (2.28 eV). The silicon-interrupted conjugation of the electron-donating and electron-accepting segments endows these materials with relative high triplet energies, good thermal and morphological stability, and bipolar transporting ability. For FIrpic-based blue PhOLEDs, the di-meta-position compounds 2 and 4 display better device performances than their di-para-position analogues 1 and 3, respectively. Device B using 2 as the host exhibits the best performance with a maximum current efficiency of 29.3 cd A−1, a maximum power efficiency of 19.8 lm W−1, and a maximum external quantum efficiency of 11.4%. Green phosphorescent devices using (ppy)2Ir(acac) as guest and 1–4 as hosts show excellent EL performances with maximum external quantum efficiencies of 18.3–22.2%. Remarkably, device H hosted by 4 still exhibits an external quantum efficiency of 19.4% at the extremely high luminance of 10 000 cd m−2. These efficiencies are significantly higher than those of blue and green control devices using mCP as host, respectively.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...