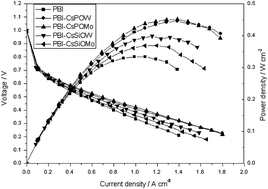
Inorganic–organic composite electrolyte membranes were fabricated from Cs substituted heteropolyacids (CsHPAs) and polybenzimidazole (PBI) for application in intermediate temperature hydrogen fuel cells. Four caesium salts of heteropolyacid, (CsXH3−XPMo12O40 (CsPOMo), CsXH3−XPW12O40 (CsPOW), CsXH4−XSiMo12O40 (CsSiOMo) and CsXH4−XSiW12O40 (CsSiOW)), were used to form composite membranes with PBI. The membranes were characterized by using SEM, FTIR and XRD. The CsHPA powders were nano-sized as shown in the XRD and SEM data. The CsHPA/PBI composite membranes loaded with H3PO4 had high conductivity, greater than that of a phosphoric acid loaded PBI membrane. The conductivity increased with an increase in the percentage of the powder in the composite. The 30% CsPOMo/PBI/H3PO4 exhibited a conductivity of 0.12 S cm−1 under anhydrous conditions, although its mechanical strength was the poorest, but still promising with a value 40 MPa. The performance of the hydrogen fuel cell with composite membranes was better than that with a phosphoric acid-doped PBI membrane under the same conditions. The CsPOMo gave the best power density of around 0.6 W cm−2 with oxygen at atmospheric pressure.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...