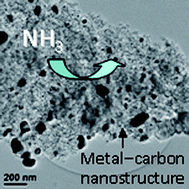
Activated carbons have long been used as substrates for the filtration of vapor-phase molecules, often with metal salts or oxides added to improve their sorption capacities for specific agents, but their real-world performance and applicability may be hindered by such factors as long-term stability and complex processing. En route to a new class of carbon-based sorbents, we have developed solid-state synthetic methods to produce bulk carbonaceous solids based on the pyrolysis of thermoset solids containing low concentrations (<1 wt%) of metal precursors (based on either Ni, Fe, or Co) that decompose in situ to catalyze the formation of a complex graphitic nanostructure. Selective combustion of residual amorphous carbon from the pyrolyzed solid generates a mesoporous network that facilitates diffusional transport of gas-phase molecules to the interior surfaces of the solid, and also converts the entrained metals to their respective metal oxide forms. We examine the ammonia-sorption properties of a series of these graphitic nanostructured compositions, and demonstrate that ammonia uptake is primarily determined by the type of residual metal oxide, with the Co-containing carbonaceous solid providing the best ammonia-sorption capacity (1.76 mol kg−1). Thermal reduction of the Co-containing material drastically decreases its ammonia-sorption capacity, showing that the oxide form (in this case Co3O4) of the entrained metal nanoparticles is most active for ammonia filtration. The effects of the carbon–oxygen functionalities on the nanostructured graphitic surfaces for ammonia sorption are also discussed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...