Analysis of biofuels production from sugar based on three criteria: Thermodynamics, bioenergetics, and product separation†
Abstract
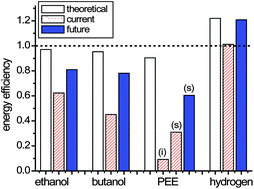
We compare the production of four biofuels -- ethanol, butanol, fatty acid ethyl ester (palmitate ethyl ester, PEE), and hydrogen from renewable carbohydrate (glucose) based on the energy-retaining efficiency that is greatly influenced by thermodyanimcs, bioenergetics, and product separation. Ethanol and butanol are produced in anaerobic fermentations; PEE is produced in semi-aerobic fermentation; hydrogen is produced by cell-free synthetic enzymatic pathway biotransformation (SyPaB), where enzymes are produced from carbohydrate by microbial fermentations. A decreasing order in theoretical energy efficiency determined by thermodynamics is hydrogen, ethanol, butanol, and PEE. Bioenergetics analysis suggests that a small fraction of carbohydrate (e.g., 5–15%) is allocated to the synthesis of cell mass in anaerobic fermentations (e.g., ethanol and butanol), a significant fraction (e.g., 20–30% or higher) has to be allocated to the synthesis of cell mass for semi-aerobic fermentations (e.g., PEE production), and a very small fraction (e.g., less than 1%) is used to produce the enzyme mixtures. A decreasing order in product separation energy is hydrogen, secreted PEE, ethanol, butanol, and intracellular PEE. Hydrogen production by SyPaB would be most appealing because its energy-retaining efficiency is ∼49% higher than ethanol, ∼55% higher than butanol, and ∼87% higher than PEE, even without considering higher hydrogen-fuel cell efficiency than those of biofuel-internal combustion engines. Our analysis suggests that it may be difficult to produce some advanced biofuels economically through aerobic fermentations due to low energy efficiency, as compared to ethanol, butanol, and hydrogen.

 Please wait while we load your content...
Please wait while we load your content...