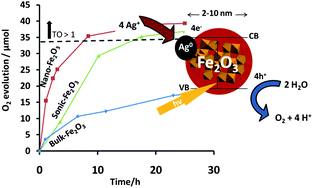
Alpha-Fe2O3 is cheap and abundant, and has a visible light indirect (phonon assisted) band gap of 2.06 eV (600 nm) due to a d–d transition, and a direct band gap at 3.3 eV (375 nm), associated with the ligand to metal charge transfer process. Here we describe results on using freely dispersed Fe2O3 nanocrystals for photocatalytic water oxidation. Three morphologies of hematite were compared, including bulk-type-α-Fe2O3 (Bulk-Fe2O3, 120 nm), ultrasonicated Bulk-Fe2O3 (Sonic-Fe2O3, 44 nm), and synthetic Fe2O3 (Nano-Fe2O3, 5.4 nm) obtained by hydrolysis of FeCl3·6H2O. According to X-ray diffraction, all phases were presented in the alpha structure type, with Nano-Fe2O3 also containing traces of β-FeOOH. UV/Vis diffuse reflectance revealed an absorption edge near 600 nm (EG = 2.06 eV) for all materials. Cyclic voltammetry gave the water oxidation overpotentials (versusNHE at pH = 7, at 1.0 mA cm−2) as η = +0.43 V for Nano-Fe2O3, η = +0.63 V for Sonic-Fe2O3, and η = +0.72 V for Bulk-Fe2O3. Under UV and visible irradiation from a 300 W Xe-arc lamp, all three materials (5.6 mg) evolved O2 from water with 20.0 mM aqueous AgNO3 as sacrificial electron acceptor. The highest rates were obtained under UV/Vis (>250 nm) irradiation with 250 μmol h−1 g−1 for Bulk-Fe2O3, 381 μmol h−1 g−1 for Sonic-Fe2O3 and 1072 μmol h−1 g−1 for Nano-Fe2O3. Turnover numbers (TON = moles O2/moles Fe2O3) were above unity for Nano-Fe2O3 (1.13) and Sonic-Fe2O3 (1.10) but not for Bulk-Fe2O3 (0.49), showing that the nanoscale morphology was beneficial for catalytic activity.

 Please wait while we load your content...
Please wait while we load your content...