Novel borothermal process for the synthesis of nanocrystalline oxides and borides of niobium†
Abstract
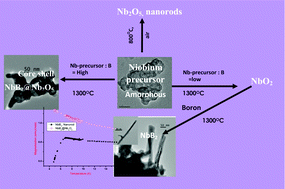
A new process has been developed for the synthesis of nanocrystalline niobium oxide and niobium diboride using an amorphous niobium precursor obtained via the solvothermal route. On varying the ratio of niobium precursor to boron and the reaction conditions, pure phases of nanostructured niobium oxides (Nb2O5, NbO2), niobium diboride (NbB2) and core–shell nanostructures of NbB2@Nb2O5 could be obtained at normal pressure and low temperature of 1300 °C compared to a temperature of 1650 °C normally used. The above borothermal process involves the in situ generation of B2O2 to yield either oxide or diboride. The niobium oxides and borides have been characterized in detail by XRD, HRTEM and EDX studies. The core–shell structure has been investigated by XPS depth profiling, EFTEM and EELS (especially to characterize the presence of boron and the shell thickness). The niobium diboride nanorods (with high aspect ratio) show a superconducting transition with the Tc of 6.4 K. In the core–shell of NbB2@Nb2O5, the superconductivity of NbB2 is masked by the niobium oxide shell and hence no superconductivity was observed. The above methodology has the benefits of realizing both oxides and borides of niobium in nanocrystalline form, in high purity and at much lower temperatures.

 Please wait while we load your content...
Please wait while we load your content...