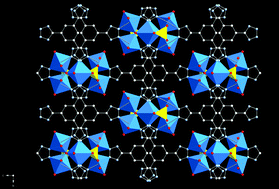
The hydrothermal chemistry of a variety of M(II)SO4 salts with the tetrazole (Ht) ligands 5,5′-(1,4-phenylene)bis(1H-tetrazole) (H2bdt), 5′,5′′-(1,1′-biphenyl)4,4′-diylbis(1H-tetrazole) (H2dbdt) and 5,5′,5′′-(1,3,5-phenylene)tris(1H-tetrazole) (H3btt) was investigated. In the case of Co(II), three phases were isolated, two of which incorporated sulfate: [Co5F2(dbdt)4(H2O)6]·2H2O (1·2H2O), [Co4(OH)2(SO4)(bdt)2(H2O)4] (2) and [Co3(OH)(SO4)(btt)(H2O)4]·3H2O (3·3H2O). The structures are three-dimensional and consist of cluster-based secondary building units: the pentanuclear {Co5F2(tetrazolate)8(H2O)6}, the tetranuclear {Co4(OH)2(SO4)2(tetrazolate)6}4−, and the trinuclear {Co3(μ3-OH)(SO4)2 (tetrazolate)3}2− for 1, 2, and 3, respectively. The Ni(II) analogue [Ni2(H0.67bdt)3]·10.5H2O (4·10.5H2O) is isomorphous with a fourth cobalt phase, the previously reported [Co2(H0.67bat)3]·20H2O and exhibits a {M(tetrazolate)3/2}∞ chain as the fundamental building block. The dense three-dimensional structure of [Zn(bdt)] (5) consists of {ZnN4}tetrahedra linked through bdt ligands bonding through N1,N3 donors at either tetrazolate terminus. In contrast to the hydrothermal synthesis of 1–5, the Cd(II) material (Me2NH2)3[Cd12Cl3(btt)8(DMF)12]·xDMF·yMeOH (DMF = dimethylformamide; x = ca. 12, y = ca. 5) was prepared in DMF/methanol. The structure is constructed from the linking of {Cd4Cl(tetrazolate)8(DMF)4}1− secondary building units to produce an open-framework material exhibiting 66.5% void volume. The magnetic properties of the Co(II) series are reflective of the structural building units.

 Please wait while we load your content...
Please wait while we load your content...