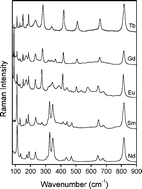
Lanthanide orthotantalates RETaO4 (RE = La, Ce, Pr, Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, and Lu) were synthesized by solid-state reaction in optimized conditions of temperature (1300 °C) and time (up to 14 h) to yield crystalline materials. The crystal structures of the obtained compounds were investigated by X-ray diffraction and Raman scattering. All the samples exhibited monoclinic structures, but with different arrangements as a function of the ionic radius of the rare-earth metal occupying the A-site. For compounds with the largest ionic radii (La–Pr), the ceramics crystallized in the monoclinic structure, P21/c (C52h, #14, Z = 4), while the compounds with intermediate ionic radii (Nd–Tb) exhibited the fergusonite M-type structure, I2/a (C62h, #15, Z = 4). Samples with the smallest ionic radii (Dy–Lu) presented the fergusonite M′-type structure, P2/a (C42h, #13, Z = 2). These last two groups of compounds exhibited phase transitions, which can be related to the processing temperature. The compounds belonging to the M-type structure are stable at high temperatures, whilst materials belonging to the M′-type structure are stable at low temperatures. The results from XRD and Raman scattering allows us to understand the differences between these two structures in terms of the vibrational-active modes assigned to each space group. Also, the Raman patterns in addition to group-theory calculations for this complete lanthanide series were reported for the first time. The experimental results are in perfect agreement with theoretical calculations: samples with P21/c (#14) structure showed 36 Raman-active modes, while both I2/a (#15) and P2/a (#13) structures exhibited 18 Raman modes.

 Please wait while we load your content...
Please wait while we load your content...