Organometallic frustrated Lewis pair chemistry
Abstract
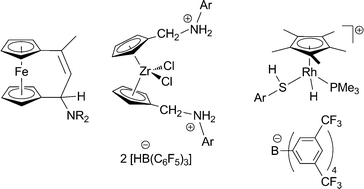
Frustrated Lewis pairs are playing an increasingly important role in organometallic chemistry. Examples are presented and discussed where organometallic systems themselves serve as the Lewis base or Lewis acid components in frustrated Lewis pair chemistry, mostly through their attached functional

 Please wait while we load your content...
Please wait while we load your content...