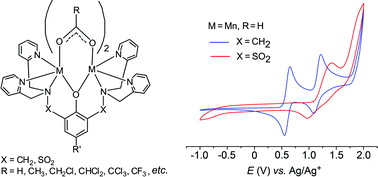
A comparison of the electrochemical properties of a series of dinuclear complexes [M2(L)(RCO2)2]+ with M = Mn or Co, L = 2,6-bis(N,N-bis-(2-pyridylmethyl)-sulfonamido)-4-methylphenolato (bpsmp−) or 2,6-bis(N,N-bis(2-pyridylmethyl)aminomethyl)-4-tert-butylphenolato (bpbp−) and R = H, CH3, CF3 or 3,4-dimethoxybenzoate demonstrates: (i) The electron-withdrawing sulfonyl groups in the backbone of bpsmp− stabilize the [M2(bpsmp)(RCO2)2]+ complexes in their MII2 oxidation state compared to their [M2(bpbp)(RCO2)2]+ analogues. Manganese complexes are stabilised by approximately 550 mV and cobalt complexes by 650 mV. (ii) The auxiliary bridging carboxylato ligands further attenuate the metal-based redox chemistry. Substitution of two acetato for two trifluoroacetato ligands shifts redox couples by 300–400 mV. Within the working potential window, reversible or quasi-reversible MIIMIII ↔ MII2 processes range from 0.31 to 1.41 V for the [Co2(L)(RCO2)2]+/2+ complexes and from 0.54 to 1.41 V for the [Mn2(L)(RCO2)2]+/2+ complexes versusAg/AgCl for E°ox(MIIMIII/MII2). The extreme limits are defined by the complexes [M2(bpbp)(CH3CO2)2]+ and [M2(bpsmp)(CF3CO2)2]+ for both metal ions. Thus, tuning the ligand field in these dinuclear complexes makes possible a range of around 0.9 V and 1.49 V for the one-electron E°ox(MIIMIII/MII2) couple of the Mn and Co complexes, respectively. The second one-electron process, MIIMIII ↔ MIII2 was also observed in some cases. The lowest potential recorded for the E°(MIII2/MIIMIII) couple was 0.63 V for [Co2(bpbp)(CH3CO2)2]2+ and the highest measurable potential was 2.23 V versusAg/AgCl for [Co2(bpsmp)(CF3CO2)2]2+.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...