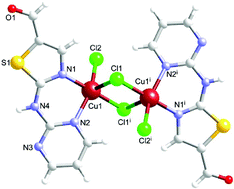
An unprecedented in situ multiple bond cleavage of S–S, S–C(sp2) and C–N in the pyrimidinedisulfide (pym2S2) ligand is observed by the reaction of CuCl2·2H2O with this ligand under solvothermal and solvothermal-microwave conditions. In this process the formation of the compound [CuII(μ-Cl)(Cl)L]2, where L represents the new ligand (L = 2-(pyrimidin-2-ylamino)-1,3-thiazole-4-carbaldehyde), is observed. This ligand has been further isolated and X-ray characterized. The similar reaction carried out under solvothermal-microwave conditions gives, in addition to the latter compound, the complex {9·[Cu(pym2S3)(μ-Cl)(Cl)]2·[Cu(pym2S2)(μ-Cl)(Cl)]2}. Coordination of a pyrimidinetrisulfide ligand (pym2S3) is reported for the first time. This work represents an illustrative example of the novel synthetic perspectives attainable via solvothermal-microwave procedures.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Please wait while we load your content...
Please wait while we load your content...