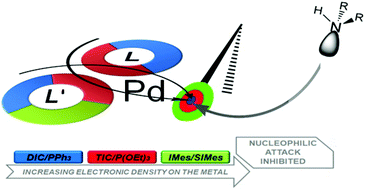
On the basis of an original protocol, we have synthesized several complexes of the type [Pd(η3-C3H3R2)(LL′)]ClO4 (R = H, Me; L, L′ = PPh3, P(OEt)3, 2,6-dimethylphenylisocyanide, t-butylisocyanide, 1,3-dimesitylimidazolidine, 1,3-dimesitylimidazol-2-ylidene). The complexes, some of which are completely new species, were fully characterized and their behaviour in solution was studied by means of 1H NMR. The reactions of the complexes bearing the symmetric allyl moiety [Pd(η3-C3H5)(LL′)]ClO4 with piperidine in the presence of the olefin dimethylfumarate were followed under kinetically controlled conditions. Formation of allyl-amine and of the palladium(0) derivatives [Pd(η2-dmfu)(LL′] was observed. The reaction rates k2 proved to be strongly dependent on the ancillary ligand nature and allowed a direct comparison among the electronic characteristics of the ligands. The reactivity trend determined appears to be mainly influenced by the capability of the ancillary ligands in transferring electron density to the metal centre and consequently on the allyl fragment.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...