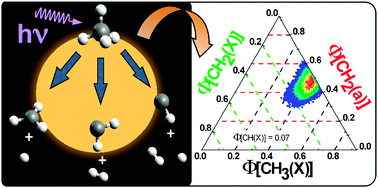
Methane photolysis has been performed at the two Vacuum UltraViolet (VUV) wavelengths, 121.6 nm and 118.2 nm, via a spectrally pure laser pump–probe technique. The first photon is used to dissociate methane (either at 121.6 nm or at 118.2 nm) and the second one is used to ionise the CH2 and CH3 fragments. The radical products, CH3(X), CH2(X), CH2(a) and C(1D), have been selectively probed by mass spectrometry. In order to quantify the fragment quantum yields from the mass spectra, the photoionisation cross sections have been carefully evaluated for the CH2 and CH3 radicals, in two steps: first, theoretical ab initio approaches have been used in order to determine the pure electronic photoionisation cross sections of CH2(X) and CH2(a), and have been rescaled with respect to the measured absolute photoionisation cross section of the CH3(X) radical. In a second step, in order to take into account the substantial vibrational energy deposited in the CH3(X) and CH2(a) radicals, the variation of their cross sections near threshold has been simulated by introducing the pertinent Franck–Condon overlaps between neutral and cation species. By adding the interpolated values of CH quantum yields measured by Rebbert and Ausloos [J. Photochem., 1972, 1, 171–176], a complete set of fragment quantum yields has been derived for the methane photodissociation at 121.6 nm, with carefully evaluated 1σ uncertainties: Φ[CH3(X)] = 0.42 ± 0.05, Φ[CH2(a)] = 0.48 ± 0.05, Φ[CH2(X)] = 0.03 ± 0.08, Φ[CH(X)] = 0.07 ± 0.01. These new data have been measured independently of the H atom fragment quantum yield, subject to many controversies in the literature. From our results, we evaluate Φ(H) = 0.55 ± 0.17 at 121.6 nm. The quantum yields for the photolysis at 118.2 nm differ notably from those measured at 121.6 nm, with a substantial production of the CH2(X) fragment: Φ[CH3(X)] = 0.26 ± 0.04, Φ[CH2(a)] = 0.17 ± 0.05, Φ[CH2(X)] = 0.48 ± 0.06, Φ[CH(X)] = 0.09 ± 0.01, Φ(H) = 1.31 ± 0.13. These new data should bring reliable and essential inputs for the photochemical models of the Titan atmosphere.

 Please wait while we load your content...
Please wait while we load your content...