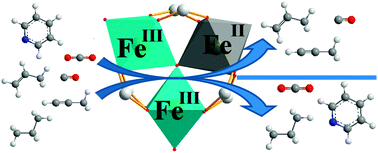
The present study illustrates the importance of the oxidation state of iron within the mesoporous iron trimesate [{Fe3O(H2O)2F0.81(OH)0.19}{C6H3(CO2)3}2] denoted MIL-100(Fe) (MIL= Material from Institut Lavoisier) during adsorption of molecules that can interact with the accessible metal sites through π-back donation. Adsorption of CO has been first followed by FTIR spectroscopy to quantify the Lewis acid sites in the dehydrated Fe(III) sample, outgassed at 150 °C, and on the partially reduced Fe(II/III), outgassed at 250 °C. The exposure of MIL-100(Fe) to CO2, propane, propene and propyne has then been studied by FTIR spectroscopy and microcalorimetry. It appears that π-back donating molecules are strongly adsorbed on reduced iron(II) sites despite the weaker Lewis acidity of cus Fe2+ sites compared to that of Fe3+ ones, as shown by pyridine adsorption.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...