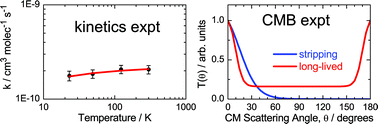
The reaction between sulfur atoms in the first electronically excited state, S(1D), and methane (CH4), has been investigated in a complementary fashion in (a) crossed-beam dynamics experiments with mass spectrometric detection and time-of-flight (TOF) analysis at two collision energies (30.4 and 33.6 kJ mol−1), (b) low temperature kinetics experiments ranging from 298 K down to 23 K, and (c) electronic structure calculations of stationary points and product energetics on the CH4S singlet potential energy surface. The rate coefficients for total loss of S(1D) are found to be very large (ca. 2 × 10−10 cm3molec−1 s−1) down to very low temperatures indicating that the overall reaction is barrier-less. Similar measurements are also performed for S(1D) + C2H2, and also for this system the rate coefficients are found to be very large (ca. 3 × 10−10 cm3molec−1 s−1) down to very low temperatures. From laboratory angular and TOF distributions at different product masses for the reaction S(1D) + CH4, it is found that the only open reaction channel at the investigated collision energies is that leading to SH + CH3. The product angular, T(θ), and translational energy, P(E′T), distributions in the center-of-mass frame are derived. The reaction dynamics are discussed in terms of two different micromechanisms: a dominant long-lived complex mechanism at small and intermediate impact parameters with a strongly polarized T(θ), and a direct pickup-type (stripping) mechanism occurring at large impact parameters with a strongly forward peaked T(θ). Interpretation of the experimental results on the S(1D) + CH4 reaction kinetics and dynamics is assisted by high-level theoretical calculations on the CH4S singlet potential energy surface. The dynamics of the SH + CH3 forming channel are compared with those of the corresponding channel (leading to OH + CH3) in the related O(1D) + CH4 reaction, previously investigated in crossed-beams in other laboratories at comparable collision energies. The possible astrophysical relevance of S(1D) reactions with hydrocarbons, especially in the chemistry of cometary comae, is discussed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...