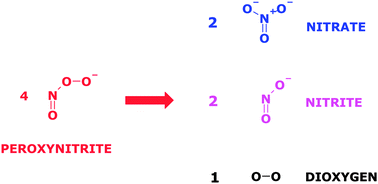
Nitric oxide (˙NO) and superoxide (O2−˙) are ubiquitous in nature. Their reaction product peroxynitrite (ONOO−) and notably its conjugated peroxynitrous acid (ONOOH) are highly unstable in aqueous phase. ONOO−/ONOOH (referred to as peroxynitrite) isomerize and decompose to NO3−, NO2− and O2. Here, we report for the first time GC-MS and HPLC methods for the analysis of peroxynitrite in aqueous solution. For GC-MS analysis peroxynitrite in alkaline solution was derivatized to a pentafluorobenzyl derivative using pentafluorobenzyl bromide. O15NOO− was synthesized from H2O2 and 15NO2− and used as internal standard. HPLC analysis was performed on stationary phases consisting of Nucleosil® 100–5C18AB or Nucleodur® C18 Gravity. The mobile phase consisted of a 10 mM aqueous solution of tetrabutylammonium hydrogen sulfate and had a pH value of 11.5. UV absorbance detection at 300 nm was used. HPLC allows simultaneous analysis of ONOO−, NO2− and NO3−. The GC-MS and HPLC methods were used to study stability, synthesis, formation from S-[15N]nitrosoglutathione (GS15NO) and KO2, and isomerization/decomposition of peroxynitrite to NO2− and NO3− in aqueous buffer.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...