A DFT study of the Karplus-type dependence of vicinal 3J(Sn–C-X-C), X=N,O,S, in organotin(iv) compounds: application to conformationally flexible systems†
Abstract
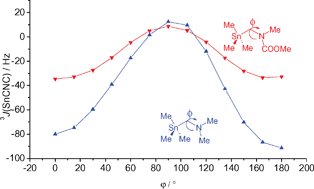
* Corresponding authors
a
Dipartimento di Chimica Inorganica e Analitica “Stanislao Cannizzaro”, Università degli Studi di Palermo, Viale delle Scienze, Parco D'Orleans II, Ed. 17, Italy
E-mail:
gcasella@unipa.it
b
Dipartimento di Chimica Fisica “Filippo Accascina”, Università degli Studi di Palermo, Viale delle Scienze, Parco D'Orleans II, Ed. 17, Italy
E-mail:
f.ferrante@unipa.it
c
Istituto per la Tecnologia delle Membrane del CNR, Unità di Padova and Dipartimento di Scienze Chimiche, Via Marzolo, Padova, Italy
E-mail:
giacomo.saielli@unipd.it
Fax: +39-049-8275239
Tel: +39-049-8275279

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
G. Casella, F. Ferrante and G. Saielli, Org. Biomol. Chem., 2010, 8, 2711 DOI: 10.1039/C000679C
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
