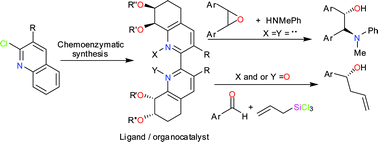
A series of enantiopure 2,2′-bipyridines have been synthesised from the corresponding cis-dihydrodiol metabolites of 2-chloroquinolines. Several of the resulting hydroxylated 2,2′-bipyridines were found to be useful chiral ligands for the asymmetric aminolysis of meso-epoxides leading to the formation of enantioenriched amino alcohols (→84% ee). N-oxide and N,N′-dioxide derivatives of these 2,2′-bipyridines, including separable atropisomers, have been synthesised and used as enantioselective organocatalysts in the asymmetric allylation of aldehydes to give allylic alcohols (→86% ee).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...