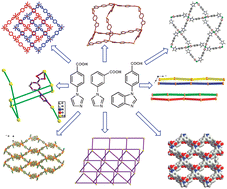
Ten coordination polymers {[Cd(L1)2]}n (1), {[Cu(L1)2]}n (2), {[Cd2(L1)2(HCOO)2(H2O)]}n (3), {[Cd2(L1)4]·3H2O}n (4), {[Cd(L1)(CH3COO)]}n (5), {[Cd(L1)2(C5H5N)]·2.5H2O}n (6), {[Zn(L2)2]}n (7), {[Cd2(L2)4]·H2O}n (8), {[Cd3(L2)6(H2O)]·2EtOH·H2O}n (9) and {[Cd(L3)2]·2DMF·4H2O}n (10), where HL1 = 4-imidazole-1-yl-benzoic acid, HL2 = 3-imidazole-1-yl-benzoic acid and HL3 = 4-benzmidazole-1-yl-benzoic acid, have been synthesized under different experimental conditions. Their structures are determined by single-crystal X-ray diffraction analyses and further characterized by IR spectra, thermogravimetric (TG) and elemental analyses. The structure of 1 is a 2D → 2D (2D = two dimensional) interpenetrating network with (4,4) grid topology while 2 has an interesting Kagome structure. Compounds 4 and 6 crystallize in 4- and 3-fold interpenetrating diamondoid frameworks respectively. Compound 7 is a layered non-interpenetrating (4,4) grid network whereas 8 has an unprecedented self-penetrating structure with 2D framework. Compound 9 is a self-penetrating 3D structure while 10 crystallizes in a non-interpenetrating (4,4) square-grid with one-dimensional (1D) channels. From these results, it is demonstrated that the structures of the coordination polymers are strongly dependent on the geometry of L1, L2 and reaction conditions. Variable temperature magnetic susceptibility study of 2 has also been performed.

 Please wait while we load your content...
Please wait while we load your content...