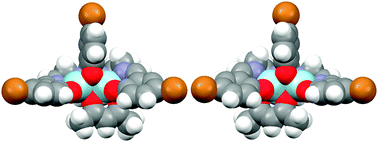
The crystal structures determined for [Zn2L1(acac)]·H2O and [Zn2L2(acac)]·H2O·MeCN·MeOH show that both compounds are chiral, although crystallising as racemates. This was not unexpected, as these complexes had been designed on the basis of the coordinating behaviour of acac in combination with 2-(2-hydroxyphenyl)-1,3-bis[4-(2-hydroxyphenyl)-3-azabut-3-enyl]-1,3-imidazolidine (H3L1) and 2-(5-bromo-2-hydroxyphenyl)-1,3-bis[4-(5-bromo-2-hydroxyphenyl)-3-azabut-3-enyl]-1,3-imidazolidine (H3L2). The reactivity of Zn(acac)2·nH2O with compartmental H3Lx (x = 1 or 2) has been investigated under different reaction conditions. Thus, depending on the temperature, the reaction with H3L1 can yield [Zn2L1(acac)]·H2O or [Zn2L1(OAc)]·H2O, whilst the interaction of Zn(acac)2·nH2O with H3L2 only gives rise to crystalline [Zn2L2(acac)]·H2O·MeCN·MeOH. This latter species loses the most volatile solvates on drying to yield [Zn2L2(acac)]·H2O. Both [Zn2Lx(acac)]·H2O complexes have been fully characterised by analytical and spectroscopic methods.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...