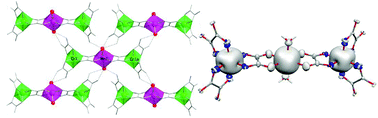
Two isostructural heterometallic trinuclear oxalato-bridged complexes of formula C4[MCr2(ox)6(H2O)2]·nH2O (C+ = 4-aminopyridinium; ox2− = oxalate dianion; M2+ = Mn2+, n = 3, 1; M2+ = Co2+, n = 3.25, 2) have been synthesized by using direct self-assembly methods combining C3[Cr(ox)3] and the chloride salts of the corresponding metal ion. The crystal structures of both compounds have been resolved by single-crystal X-ray diffraction. They crystallize in the C2/c space group [a = 11.5113(15) Å, b = 20.250(3) Å, c = 21.810(4) Å, β = 100.447(10)°, V = 5161.6(3) Å3, and Z = 4 for 1, and a = 11.4334(16) Å, b = 20.243(2) Å, c = 21.805(3) Å, β = 101.113(9)°, V = 4951.9(11) Å3, and Z = 4 for 2]. The structures of 1 and 2 consist of discrete linear [MCr2(ox)6]4− bimetallic trinuclear units, pyridinium cations and crystallization water molecules. The linear trinuclear unit is built from a central trans-diaquametal(II), linked to two Cr(ox)3]3− entities by oxalate bridges. One of the oxalate ions is coordinated to the central metal ion whereas the other two oxalate ligands are non-bridging. In the crystal, intermolecular hydrogen bonds involving oxalate ligands, water molecules and pyridinium cations, build a complex three-dimensional network. Variable-temperature magnetic susceptibility measurements for 1 and 2 indicate a weak ferromagnetic interaction (J = +1.16 and +2.62/+2.70 cm−1 for 1 and 2, respectively) between the two terminal CrIII (SCr = 3/2) and the central high-spin MnII (SMn = 5/2) and CoII (SCo = 3/2) ions. The nature and the amplitude of the exchange interaction are rationalized using DFT calculations and orbital interpretations.

 Please wait while we load your content...
Please wait while we load your content...