Influence of OHgroups on charge transport across organic–organic interfaces: a systematic approach employing an “ideal” device
Abstract
The charge
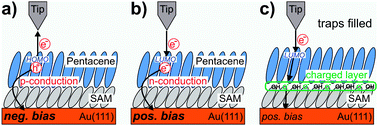
- This article is part of the themed collection: Interfacial Systems Chemistry: Out of the Vacuum, Through the Liquid, Into the cell

 Please wait while we load your content...
Please wait while we load your content...