Derivatives of cysteine related to the thiosulfate metabolism of sulfur bacteria by the multi-enzyme complex “Sox”—studied by B3LYP-PCM and G3X(MP2) calculations†
Abstract
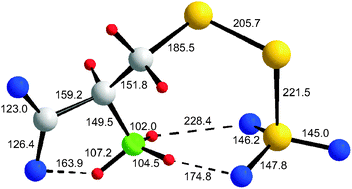
Certain sulfur bacteria oxidize thiosulfate enzymatically to sulfate, and derivatives of the amino acid cysteine play an important role as intermediates in this process. Since some of the proposed intermediates have so far been of hypothetical nature, we have investigated the structures and thermodynamic properties of more than 60 related derivatives of cysteine (CysH) by high-level quantum chemical calculations both in the gas phase and in a polarizable continuum using the PCM method to simulate an aqueous solution. Most of these molecules and anions were studied for the first time. Especially for the smaller species several conformational isomers of similar energy were identified; their relative stabilities are mainly determined by intramolecular hydrogen bonds. In contrast to the thiolate ion [Cys]−, the gaseous anions [CysS]−, [CysSO2]−, [CysSO3]− and [CysSSO3]− are most stable as zwitterions containing an NH3 rather than an NH2 group. This result also holds for the polarizable continuum. On the other hand, the related neutral molecules CysH, CysSH and CysSO2H are predicted to exist as NH2 derivatives rather than zwitterions in the gas phase and this connectivity is predicted for CysH and CysSH also in the polarizable continuum. A model molecule of composition C4H7N2O2SH (abbreviated as RSH) simulating the structural environment of a cysteine residue within the peptide chain near the corresponding reaction center of the thiosulfate oxidizing enzyme complex “Sox” was used to elucidate the geometry of the proposed reaction intermediates as well as their thermodynamic properties. In the polarizable phase, the S-sulfonate ions [CysSO3]− and [RSSO3]− are predicted to react exothermically with water to the corresponding thiol and hydrogensulfate ions. These results support the proposed mechanism for enzymatic thiosulfate metabolism. Sulfur dioxide and hydrogensulfite anions are predicted to react exothermically and exergonically with thiolate and persulfide anions to give the corresponding S-sulfinate species [RSSO2]− and [RSSSO2]−, respectively. The latter ions help to explain the inhibition of certain thiolate based enzymes by aqueous sulfite, disulfite and dithionite anions in sulfur oxidizing microorganisms.

 Please wait while we load your content...
Please wait while we load your content...