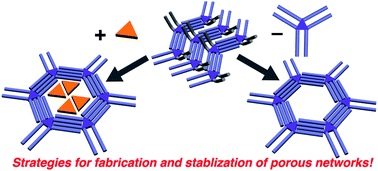
Surface-confined porous molecular networks can be formed by self-assembly at the liquid–solid interface by controlling the subtle interplay between molecules, solvent and substrate. Typically, directional interactions such as hydrogen bonding or metal–ligand interactions are at play in defining the topology of the networks. However, porous molecular networks maintained only by van der Waals interactions are at the focus in this Highlight. The liquid–solid interface at atomically flat conductive substrates such as graphite is an excellent medium to support the self-assembly of alkylated molecules. Molecular symmetry defines the shape of the pores, ranging from diamond to honeycomb and Kagomé. Solvent often plays an important role and sometimes several porous and non-porous structures are formed. The solute concentration is a key factor too, the formation of porous patterns is favoured at low concentrations. These pores, with diameters ranging from about three nanometres to more than seven nanometres, can be filled with guest molecules. In some cases, these porous topologies can only be formed upon addition of template molecules, which direct the structural transformation from a high-density to a low-density, i.e. porous, phase. The template molecules stabilize the pores. Following appropriate design principles, multicomponent networks can be formed, containing up to four different organic molecules.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...