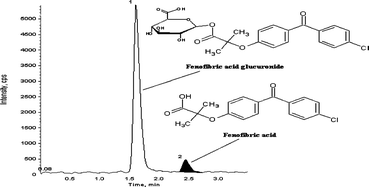
This is the first publication on a validated bioanalytical method for estimation of fenofibric acid in human EDTA plasma that chromatographically resolves its acyl glucuronide. An API 3000 mass spectrometer was employed in this method, and fenofibric acid-d6 served as the internal standard. Sample preparation involved solid phase extraction (SPE) using a polymer based, hydrophilic-lipophilic balanced cartridges and samples were chromatographed on a Discovery C18, (4.6 × 50 mm, 5 μm) column using a mobile phase of 0.2% formic acid solution/acetonitrile (35 : 65, v/v). Negative mass transitions (m/z) of fenofibric acid and fenofibric acid-d6 were detected in multiple reactions monitoring (MRM) mode at 317.1 → 230.9 and 322.9 → 230.8, respectively. The method was validated over a concentration range of 0.150 μg mL−1 to 20.383 μg mL−1. Intra- and inter-run imprecision of fenofibric acid assay at four concentration levels was below 2.5%, and inaccuracy was within ±2.8%. Analytical recoveries for fenofibric acid and internal standard were 73.8–75.4%, and 85.9%, respectively. All other validation parameters were within acceptable limits. The back conversion of fenofibric acid from its acyl glucuronide was minimized by processing samples in an ice cold water bath under low light.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...