13C NMR spectroscopy for determining the acylglycerol positional composition of lampante olive oils. Chemical shift assignments and their dependence on sample concentration
Abstract
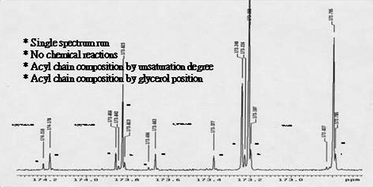
Two Lampante olive oils with free acidity expressed as
* Corresponding authors
a
CRA Centro per l'Olivicoltura e l'Industria Olearia, Sede Scientifica di Città S. Angelo, Viale Petruzzi n. 75, Città S. Angelo, Pescara, Italy
E-mail:
g.vlahov@tiscali.it
Fax: +39(0) 85 959518
Tel: +39 (0) 85 95212
Two Lampante olive oils with free acidity expressed as

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
G. Vlahov, A. A. Giuliani and P. Del Re, Anal. Methods, 2010, 2, 916 DOI: 10.1039/C0AY00028K
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
