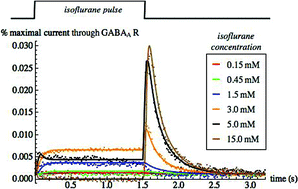
The time- and concentration-dependence of agonist-induced ion currents through postsynaptic receptors is often remarkably complex, as is the modulation of these currents by other solutes such as anesthetics. Traditionally, kinetic models have been developed that involve agonist binding and conformational transitions among a manifold of protein conformational states engineered to reproduce the complexity of the electrophysiological results. In the present work, an alternative model is proposed that minimizes the number of conformational states while additionally incorporating effects of adsorption of agonist and nonbinding compounds such as anesthetics to the bilayer in which these intrinsic membrane proteins are embedded. Adsorption of these aqueous solutes alters bilayer physical properties, which in turn can distort the protein conformational free energy landscape, and thus alter the rate constants of protein conformational transitions. The complexity of the predicted ion currents then arises from the time-dependence of solute adsorption, resulting in strongly time-dependent transition rate “constants”. If only nonbinding solutes are present, the model simplifies considerably. Best fits with respect to a small set of parameters of the predicted current traces are found to be in excellent agreement with previously measured currents in GABAA receptors induced by a broad range of supraclinical concentrations of isoflurane and sevoflurane.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...