Chemical disorder and spin crossover in a mixed ethanol–2-propanol solvate of FeII tris(2-picolylamine) dichloride†
Abstract
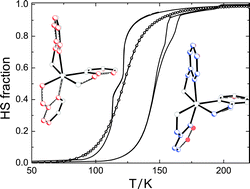
We compare the macroscopic and magnetic properties of three spin crossover compounds, [FeII(2-pic)3]Cl2·EtOH, [FeII(2-pic)3]Cl2·2-PrOH and [FeII(2-pic)3]Cl2·(EtOH)0.744·(2-PrOH)0.256 (pic =
- This article is part of the themed collection: 3rd International Symposium on Molecular Materials (MOLMAT)

 Please wait while we load your content...
Please wait while we load your content...