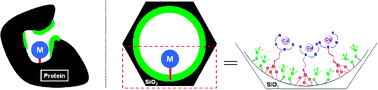
A bio-inspired synthesis of a silica grafted polydentate copper(II) complex is developed following the structural concept of metalloproteins where well defined metal ion coordination state, hydrophobic environment and confined space are present. Mesostructured porous silica of MCM-41 type replaces the proteic matrix while the pore surface is engineered according to a molecular stencil patterning technique combining both partial hydrophobization and site isolation in order to mimic the enzymatic cavity. The overall five-step synthesis includes the sol–gel formation of the silica matrix followed by partial removal of the structure directing agent and, sequential surface chemical modifications. This new methodology is illustrated here using trimethylsilyl functions to dilute bromopropylsilyl tripod tethers that undergo, directly in the pores, in a subsequent step nucleophilic substitution by a tetradentate ligand N,N′-bis(2-pyridinylmethyl)ethane-1,2-diamine (L42). The metallation of the grafted ligand is obtained in the final step by merely contacting the solid with copper(II) chloride or triflate ethanolic solutions. Different techniques such as powder XRD, N2 adsorption–desorption, elemental analysis, IR, XPS, EPR and EXAFS were combined together with an emphasis on quantification to reach a quasi-molecular description at each functionalisation step of the internal surface of the materials.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...