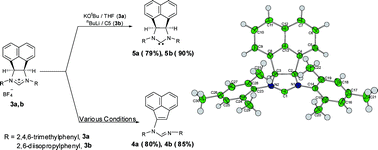
Stable annulated diaminocarbene ligands 7,9-bis(2,4,6-trimethylphenyl)-6b,9a-dihydroace naphtha[1,2-d]imidazolin-2-ylidene and 7,9-bis(2,6-diisopropylphenyl)-6b,9a-dihydroacenaphtho[1,2-d]imidazolin-2-ylidene; designated as (BIAN-SIMes, 5a,) and (BIAN-SIPr, 5b), respectively, have been prepared. The base dependent decomposition of imidazolinium salts via ring opening at the backbone was also observed. The corresponding rhodium(I) and iridum(I) complexes (η4-1,5-COD)M(BIAN-SIMes)Cl and (η4-1,5-COD)M(BIAN-SIPr)Cl; M= Rh (6a, 6b) and Ir (7a, 7b) have been synthesised by the reaction of free carbene with [M(η4-1,5-COD)(μ-Cl)]2; where M= Rh, Ir. The cationic Ir(I) complexes [(η4-1,5-COD)Ir(BIAN-SIMes)Py]BF48a and [(η4-1,5-COD)Ir(BIAN -SIPr)Py]PF68b have also been synthesised. Compounds 4b, 5a, 6a, 6b, 7b and 8b have been structurally characterised. The catalytic activities for the rhodium(I) complexes 6a and 6b were evaluated for the hydroformylation of 1-octene.

 Please wait while we load your content...
Please wait while we load your content...