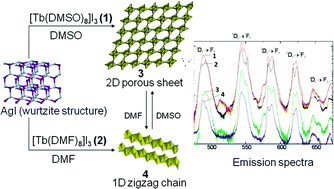
Octakis solvated terbium iodide complexes [Tb(DMSO)8]I3 (1) and [Tb(DMF)8]I3 (2) (DMSO = dimethylsulfoxide; DMF = dimethylformamide) were used as synthons to construct novel iodoargentate fabrics. Reaction of 1 with 6 equivalents of AgI in the presence of NH4I produced a novel framework compound [Tb(DMSO)8]2[Ag2(μ-I)3I2][Ag5(μ3-I)4(μ-I)4]2∞ (3) involving alternate layers of discrete [Tb(DMSO)8]3+ cation, [Ag2I5]3− anion and a 2D microporous sheet of the composition [Ag5I8]3−, whereas similar reaction of 2 yielded [Tb(DMF)8][Ag6(μ4-I)2(μ3-I)2(μ-I)5]1∞ (4) with a 1D zigzag structure. The two structural forms are completely inter-convertible on mutual exchange of DMSO and DMF ligands, the partial substitution of the former ligand in 3 yielding a mixed-ligand complex [Tb(DMF)6.7(DMSO)1.3][Ag6(μ4-I)2(μ3-I)2(μ-I)5]1∞ (5). Single crystal X-ray structures are reported for all the complexes. Thermo-gravimetric analyses of these complexes show that thermal stability decreases in the order 4 > 2 > 3 > 1, whereas the diffuse reflectance spectra of 3 and 4 exhibit an optical band gap of about 3.7 eV, thus revealing the insulator nature of these hybrid iodoargentates. Compared to solvated terbium iodide complexes 1 and 2, the high energy transitions in the excitation spectra of the iodoargentate complexes 3 and 4 are quenched by a process which can best be attributed to the auto-ionization of the carriers in the above materials.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...